Challenges when Assessing Hydrocarbon Degradation
via Isotopic Fractionation
By:
John Kessler
University of Rochester
| When: | Friday, October 12, 2012, 10:30 a.m. to 11:30 a.m. Join us for coffee beginning at 10:00 a.m. |
| Where: | Seminar Conference Room, 10100 Burnet Road, Bldg 196-ROC, Austin, Texas 78758 |
| Host: | Peter Flemings, UTIG |
Click for a Live Broadcast.
Abstract
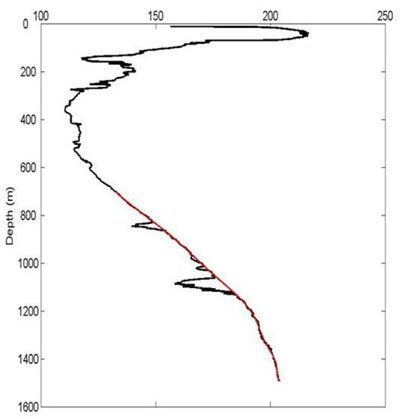
A fundamental question when assessing the fate of a chemical perturbation in a natural system is "how much of this perturbation was biodegraded vs. dispersed and diluted?" Measurements of the stable isotopes of the chemical perturbation help to answer this question due to Rayleigh fractionation, a kinetic process that occurs in this case during biodegradation but not dilution.
However, this process is not straight-forward when microorganisms grow up in response to an increased food source. Laboratory measurements and models produce conflicting results on how microbes isotopically fractionate chemicals during different stages of microbial growth. The answer to this debate may be revealed by the Deepwater Horizon incident where microbes bloomed in response to the increased hydrocarbon abundances following the well blowout.
After introducing the original debate, this talk will present the concentration, rate, and isotope data related to the Deepwater Horizon incident to provide insights into this fundamental isotopic process and ultimately lead to a more quantitative framework for assessing the fate of future releases.